 SynLube™
Lube‑4‑Life®
SynLube™
Lube‑4‑Life®
 SynLube™
Lube‑4‑Life®
SynLube™
Lube‑4‑Life®
Some of the most common questions that we receive about SynLube™ Lube‑4‑Life® products are indicative of the fact that majority of our potential customers have no real idea how automotive engine functions!
This article is intended to give some insight in to the how and why Engine works.
This article will explain how Conventional Gasoline or Diesel Piston Engine
works, and where the FUEL that you put into your tank goes.
Once you understand engine operation, utilization of the FUEL you will have
greater appreciation for function of a lubricant in the operation of all
Engines.
Then you will be able to understand concepts like Sol Lubrication and how it differs from conventional Liquid Oil Lubrication.
Current Price of Fuel in USA
| Gasoline Prices |
Every engine in order to operate needs all of following:
We will now discus each of these in detail.
Where does the Fuel go?
Have you ever given any thought to where does the fuel go, or what happens to it in your car?
Although there are millions of drivers and thousands of mechanics, only small fraction of them knows or can answer this question about Fuel.
Most will "sort of" guess and be perhaps close in their belief to the actual reality.
There however is no reason why you should not know or understand what "exactly" happens to it.
First of all you probably already know that when there is no Gasoline in the fuel tank, that the car stops!
Therefore fuel is necessary for engine operation.
Gasoline also known in some parts of the World as Petrol or Benzin contains virtually hundreds of different chemicals, the major proportion of which are known as Hydrocarbons.
Hydrocarbons are chemical compounds consisting solely of Hydrogen and Carbon atoms.
But no matter how much Gasoline you have in your fuel tank, the potential energy that is stored in it must be first released before it can be harnessed to propel a vehicle.
The second essential component necessary for this energy release is:
This second component is needed to make the car go.
Perhaps you may know from experience that if the Air Filter is dirty or clogged that the car does not run as well or is nearly impossible to start.
Likewise, if you plug up the Air Intake with anything, the engine will stop.
Therefore, Air is also necessary for engine operation.
The Air that we breathe and the Air that is also used in the automobile engine is a mixture (by volume) of following gases:
More precise composition is shown in table below:
However, only the Oxygen in the Air supports the
combustion process of Gasoline or Diesel
Fuel
Combustion is a rapid burning (oxidation) of fuel and it generates vast amount of heat in a very short time.
Combustion process occurs only if there is proper ratio of Air and Fuel.
This proper or ideal ratio is called "Stoichiometric" Air/Fuel Ratio and is scientifically expressed by Greek letter Lambda = λ.
If there is exactly correct amount of Air and Fuel in the
mix the Lambda is equal to one
( λ = 1 ).
If there is more Fuel than this "ideal" ratio requires, or in other words there is an excess of fuel, the Air/Fuel mix is said to be "richer" or "rich" and Lambda is less than one ( λ < 1 ).
If there is not enough Fuel that the "ideal" ratio requires, or insufficient fuel, or in other words that mean the same thing an Air excess, the Air/Fuel mix is said to be "leaner" or "lean" and the Lambda is more than one ( λ > 1 ).
In case of Gasoline or Diesel Fuel the "Stoichiometric" Air/Fuel ratio is 14.5 : 1 by Weight.
That is one pound of Gasoline requires fourteen and half pounds of Air to burn completely.
Of course it is pretty difficult to imagine 14.5 pounds of Air, so
imagine this:
one gallon of Gasoline requires as much as 1,200 cubic feet or
approximately 9,000 gallons of Air to burn
completely.
The "richer" (excess Fuel) or "leaner" (excess Air) the Air/Fuel mixture is, the harder it is to ignite and the slower is the combustion process. At the "upper" (too much Fuel) and "lower" (too much Air) ignition limits, the mixture is no longer ignitable and the engine stops.
If the Air/Fuel mixture is ideal the automotive engine produces only Carbon Dioxide (gas), Water vapor (steam) and heat as a result of its "complete" combustion process.
The chemical reactions that occur during combustion of any hydrocarbon fuel are as follows:
For Carbon component: C + O2 = CO2
For Hydrogen component: 2H2 + O2 = 2H2O
Unfortunately due to rapid revolution of the typical engine and less then ideal mixing of Fuel and Air, the actual combustion in typical engine is far from perfect. As a result we have Air Pollution, Smog, and laws that insist on "clean" exhaust from modern vehicles.
Since combustion and complete burning in conventional engines is difficult to achieve, engineers have for over 40 years resorted to "after-treatment", hence we now have catalytic converters, unleaded fuel, low sulfur fuel, etc.
In an automotive engine it is the heat that is important. It is the heat that is used to generate useful power. The heat in turn is converted in the engine to mechanical power output, which is then used to propel the car.
Since it is currently very difficult to precisely monitor the Air/Fuel mixture for a proper ratio "before" it enters the engine, the Air/Fuel ratio is monitored after the combustion process in the exhaust system by monitoring the Oxygen content in the exhaust stream.
The device that is used for this monitoring is a special electrode that generates low voltage in 0.05 to 1.0 Volt range. This device is called "Exhaust Oxygen Sensor" or "Lambda Sensor".
If there is exactly correct amount of Air and Fuel in the
mix the Lambda is equal to one
( l = 1 ) and the sensor output is in
0.45 to 0.55 Volt range.
If there is more Fuel than this "ideal" ratio requires, or in other words there is an excess of fuel, the Air/Fuel mix is said to be "richer" or "rich" and Lambda is less than one (l < 1 ) the sensor output is higher and up to 1 Volt.
If there is not enough Fuel that the "ideal" ratio requires, or insufficient fuel, or in other words that mean the same thing an Air excess, the Air/Fuel mix is said to be "leaner" or "lean" and the Lambda is more than one ( l > 1 ) and the sensor output is lower and can be as low as 0.05 Volt.
This sensor output is used in modern computer controlled engines with fuel injection to time the duration of fuel injection and thus continuously adjust the Air/Fuel mixture as the engine operates up to several times per second.
This in turn minimizes the fuel consumption and minimizes the exhaust emissions of pollutants such as CO (Carbon-Monoxide), HC (Unburned Hydrocarbons) and NOx (Nitrides of Oxygen = major smog forming component).
The spark ignition four stroke engine was first devised by Nikolaus Otto in Germany in 1876, it is the type of power plant that is used in majority of motor vehicles today.
The Otto engine is an internal combustion engine, which converts the chemical energy contained in the fuel (usually Gasoline) into a kinetic energy by burning it (rapid oxidation).
The energy conversion is carried out by combustion of the hydrocarbons in the Fuel. There is one important thing to understand about the hydrocarbon fuel, such as Gasoline, in reference to combustion. That is, it is the energy stored in the chemical bond within the hydrocarbons themselves that is released when these compounds burn in the presence of Oxygen inside the combustion chamber. This released energy is Heat.
The internal combustion engine is therefore a Heat Engine, and it is the Heat inside the engine that is converted into a useful kinetic rotary motion by mechanical means.
The combustion process in the Otto engine is initiated by electric spark within the combustion chamber after the Air/Fuel mixture was compressed.
The image below shows some key components of the Spark Ignition Engine:
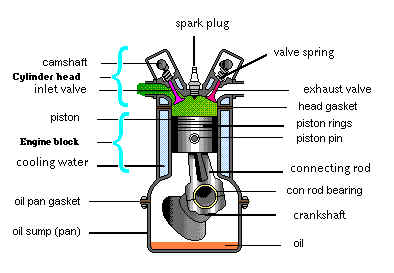
In the spark ignition engine an Air/Fuel mixture is usually formed
outside the combustion chamber. This mixture is generated in a
Carburetor or by means of Fuel Injection, but in either case
the final Air/Fuel mixture is fed into the Cylinder, through
the Intake, past the Inlet Valve.
The mixture is then Compressed and subsequently Ignited by
the Spark Plug.
The combustion of ignitable Air/Fuel mixture is initiated (Ignited) by an
Electric Spark and burnt inside a working Cylinder.
When any Gas is heated, it expands in volume, or if the volume is limited it undergoes increase in pressure.
The Piston Engine utilizes both of these Physical phenomena to convert the energy stored in fuel into a mechanical motion.
During combustion Heat given off increases the pressure of
the pre-compressed gasses.
This after-combustion pressure is typically 400 to 700 PSI, which is much
higher than the pre-combustion pressure of 95 to 155 PSI. (PSI = Pounds per
Square Inch)
This high pressure produces mechanical work by forcing the Piston down and via Pin and Connecting Rod causes the Crankshaft to turn.
After each Power Stroke the burnt gases are expelled by the Piston during upward motion and discharged into the atmosphere past the Outlet Valve (Exhaust Valve) through Exhaust tract.
The exchange of gas in the 4-stroke spark ignition engine is controlled by Valves, which open or close the Inlet and Outlet ports of the Cylinder, depending on the position of the Piston and the Crankshaft. The opening and closing period of the Valves is in turn operated by Camshaft, which turns at one half the speed of Crankshaft.
The four strokes of a working cycle are:
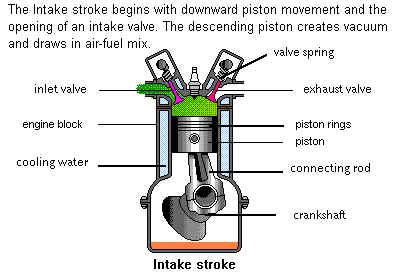
The downward movement of the Piston increases the volume of the combustion chamber, enabling a fresh Air/Fuel mixture to be sucked in past the open Inlet Valve into the Cylinder.
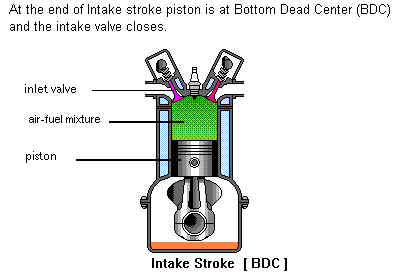
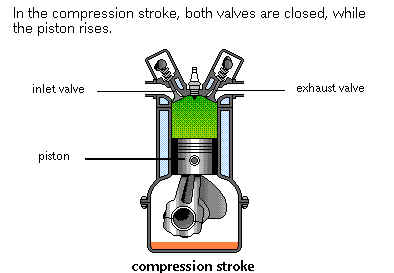
The upward moving Piston reduces the volume of the combustion chamber thereby compressing the Air/Fuel mixture. The compression factor is approximately from 6 to 14, according to the type and design of the engine. The final compression pressure is from 85 to 190 PSI.
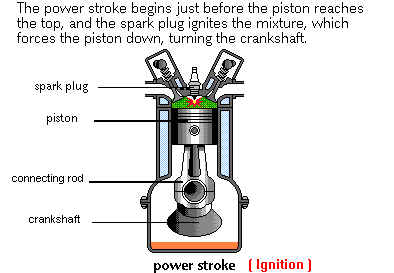
The compressed Air/Fuel mixture is Ignited by the Electric
Spark at the Spark Plug electrode gap. As the mixture is
rapidly burnt its temperature also rapidly increases, typically to up to
4,000°F (2,200°C).
As a result the pressure in the Cylinder rises to between 400
to 600 PSI (27 to 41 bar).
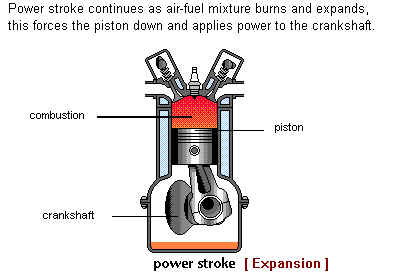
The pressure of the combustion gasses drives the Piston downwards in the Cylinder and by means of the Con Rod (Connecting Rod), produces rotary movement of the Crankshaft.
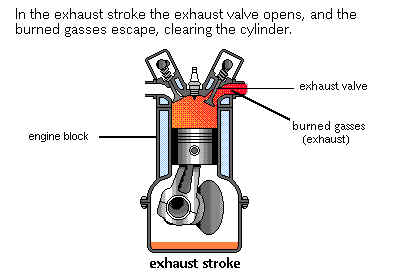
The upward moving Piston reduces the volume of the combustion chamber, whereby the burnt gases (exhaust) are expelled through the open Outlet Valve and through the Exhaust tract to the atmosphere.
In practice on most engines an Exhaust Muffler or a Silencer is used to dissipate a high kinetic energy that is present in the exhaust gases and which would otherwise cause uncomfortably loud noise.
In other applications the exhaust gasses are expanded in a high-speed turbine (Turbocharger) and the recovered energy is used for supercharging of the Intake with Air/Fuel mixture at higher than atmospheric pressure (Turbo Boost). This method is frequently used in high performance cars and high altitude piston engine aircraft.
The stroke cycle repeats itself after the Fourth stroke. In the actual cycles of the internal combustion engine the opening times of the Valves are synchronized to the movement of the Piston and to the rotation of the Crankshaft by the use of Camshaft(s), Push Rods and or Rockers.
Each cycle of the four strokes employs two rotations of the Crankshaft and one rotation of the Camshaft(s).
Typical engine idles at about 900 RPM, that is 450 cycles occur in every
minute of operation.
In actual operation most engines operate in 2,000 to 4,500 RPM range. On the
other hand,
a high performance racing engines develop peak power at between 10,000 to up to
15,000 RPM.
The efficiency of the engine depends to a large extend upon the following criteria:
The higher the Compression Ratio or the pre-compression pressure, then the higher is the thermal efficiency of the internal combustion engine. This results in a better fuel usage and more power is developed while less fuel is consumed. The maximum compression is however limited by the Octane Rating of the Gasoline that will be used. The higher the Octane Rating the higher the compression can be.
Unfortunately, higher Octane Gasoline costs more to produce than low Octane Gasoline. Therefore the increase in fuel efficiency can be offset by increase in fuel costs.
The Compression Ratio is based on the mechanical design of the engine and is expressed as:
e = (Vh + Vc) / Vc
Where:
e = Compression Ratio
Vh = Cylinder swept Volume
Vc = Combustion space Volume of Cylinder
Even more important than Compression Ratio is the actual pre-compression pressure also called Final Compression Pressure. Although its value can be also described and figured out mathematically, it is always substantially less than the mathematical result. The actual Final Compression Pressure can be reliably obtained only by a measurement with a special tool, the Compression Tester.
It is however important to know what the Final Compression Pressure should be for the particular engine. This specification can be usually found in a "Shop Manual" for the particular engine. The difference between the measured and specified values for the Final Compression Pressure determines the "Sealing Quality" of the combustion chamber.
The quality of the combustion chamber sealing by means of the Piston Rings and the Valves is a measure of the condition of the engine. Lubricant can also affect the quality of the sealing between the Rings and the Cylinder bore.
When the Final Compression Pressure is too high on a used engine, it usually means that the combustion chamber and the piston crown have excessive amounts of carbon deposits that have been formed due to any of the following:
If the Final Compression Pressure is too low on a used engine, it usually means that the engine has any of the following problems:
For the quality of the combustion process it is of prime importance that the fuel mixes intimately with the air, so that it can be burnt as completely as possible. It is important that the flame front progresses spatially and in regular form during the power stroke, until the whole mixture has been burnt. The combustion process is considerably influenced by the point in the combustion chamber at which the mixture is ignited, and by the mixture ratio as well as the manner in which it is fed into the combustion chamber.
Combustion is optimal and the efficiency of the engine is at its best when the residual gases contain no unburned fuel and as little of Oxygen as possible. The Hydrocarbons are broken up during the combustion into their constituent parts, they are Hydrogen and Carbon. On complete combustion the Carbon and Hydrogen burn to form Carbon Dioxide and Water vapor. When the combustion is incomplete the exhaust gases also contain other undesirable constituents.
The Specific Fuel Consumption of an engine is defined as the amount of energy produced per given amount of fuel consumed in the combustion process. The amount of fuel is quoted in grams or kilograms and the amount of energy produced in Kilo-Watt-Hours or Horsepower per hour.
Internal combustion engines can consume as little as 300 grams per kWh or as much as 1,200 grams per kWh.
In general the Specific Fuel Consumption is at its greatest (least efficient) when the engine is subjected to low loads, such as idle. This is because the ratio between the idling losses (due to friction, leaks, and poor fuel distribution) and the brake horsepower is the most unfavorable.
Most engines have the lowest Specific Fuel Consumption at three-quarter load, which is at 75% of the maximum power output and at about 2,000 RPM.
The Specific Fuel Consumption of engine is for the most part dependent on the mixture ratio of the Air/Fuel mixture. Consumption is at its lowest with an Air/Fuel Ratio of approximately 15 pounds of Air to one pound of Fuel. This means that 10,000 gallons of Air are needed to burn one gallon of Gasoline.
The mechanical design of the internal combustion engine has not changed since its conception in 1876, mainly because it works. The problem is, that it has been invented long before there was thorough understanding of thermodynamics or of the chemical reactions during combustion process. Further cheap and plentiful fuel -- Gasoline was easily available and until few years ago there was no concern with conservation or pollution.
As a result the internal combustion engine is an energy efficiency dinosaur that refuses to die.
To give you some idea why that is so, let's consider this:
Gasoline contains about 42 to 43.5 Mega-Joules of energy in one Kilogram that is equal to about 18,060 to 18,705 Btu per pound.
U.S. Gallon of Regular Gasoline (87 Octane) is approximately 132x106 joules of energy, equivalent to 125,000 BTU or 36,650 watt-hours.
However, Gasoline in USA contains 10% of Ethanol, as well as other Chemical
Additives.
This is because by Government decree it is required for two reasons:
As a result the Pump Gasoline in USA has the energy equivalent of ranging from low of 32.6 kWh to high of 35 kWh, EPA uses 33.7 kWh as "Gasoline Energy Equivalent" for it's calculations.
The pie chart below shows where all that energy that is available in Gasoline goes:
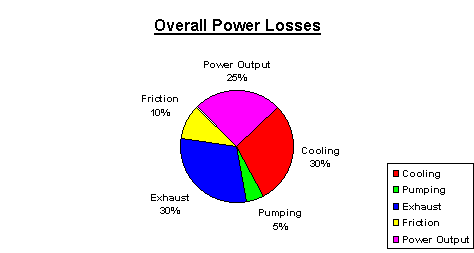
It may amaze you to know that only a quarter of the energy that is available in the Gasoline is converted into useful kinetic energy at the crankshaft. The rest or 75% is dissipated into the environment mostly as Heat.
With millions of motor vehicles in operation at any given moment on the Earth, just imagine what that does for "Global Warming"!
The 30% of the energy that is available in the Gasoline is used to Heat up the ambient Air, either directly, as is the case with Air-Cooled Engines, or indirectly as is the case with Liquid or Water Cooled Engines.
The reason why this waste is necessary is because the internal combustion engine is a Heat Engine, that is, Heat is produced by it. If the Heat can not be removed as quickly as it is produced the materials (Metals and Plastics) from which the engine is made would literally melt.
On highly efficient engine designs the losses through cooling system may be reduced to as low as 20%. This is achieved through the use of much more expensive materials that can be safely operated at higher temperatures without damage. The disadvantage of such designs is their high cost as compared to conventional designs and also the higher operating temperature causes the lubricant to deteriorate much more rapidly.
For every increase of 8°C to 10°C (12°F to 18°F) the oxidation rate of lubricant doubles and therefore the life of lubricant is cut in half. As a result motor oil must be changed twice as often, or else much more expensive synthetic lubricant must be used.
This is where there exists a great potential for new engine designs that are specifically designed to use SynLube™ Lube‑4‑Life® as engine lubricant -- they can be safely operated at continuous temperatures of up to 350 °F (175°C). By contrast conventional petroleum lubricants cause problems at temperatures exceeding 195°F (90°C). The increase in operating temperatures of specially designed engines can reduce the Cooling Losses from 30% down to 10%. This would almost double the power that is available for propulsion.
Just as is the case with Cooling, another 30% of energy is lost as Heat in the Exhaust gases. This is another area of great waste.
In some more racy engine designs as much as 35% of energy is lost in the exhaust and it is quite often enough to make the exhaust pipes glow red hot under full power output.
Some of this energy can be recovered with the use of exhaust driven turbo, output of which is usually used to drive another turbine that is used to supercharge the intake Air/Fuel mix.
There is no reason, other than cost of the equipment, why this waste heat
energy could not be used to drive electric generators, air conditioner
compressors or other accessories either directly or through the use of mini
"Steam Engines".
However, no manufacturer has yet designed any production vehicle or engine
system that fully utilizes all this Exhaust energy.
Pumping Losses
They amount to 5% of the energy that is available in the Gasoline. They comprise of the power that is used to suck the Air/Fuel mix into the engine, the power that is used to Compress the Air/Fuel mix, the power that is used to circulate the coolant and the power that is used to pump the motor oil in the engine.
Not much can be done about Pumping Losses, they are all important to the engine operation. The power that is used to circulate engine coolant can be eliminated by changing the engine design from Water Cooled to Air-Cooled.
Some of the Oil Pump Pumping Loss can be reduced by the use of lower viscosity lubricants and especially with the use of lubricants with high Viscosity Index.
In a typical automobile the oil pump circulates about 4 gallons of oil every minute while pressurizing it to at least 40 PSI. At temperature of 200° F typical SAE 30 Petroleum Motor Oil has Kinematic Viscosity of 10 Centistokes (cP). With Oil this calculates to be equivalent to a power consumption of .12 HP (90 Watts).
However with SynLube™ Lube‑4‑Life® the Kinematic Viscosity will be at least 15 Centistokes (this is beneficial because higher viscosity allows for better oil film formation and reduction of wear), however due to different physical properties (Specific Gravity, Viscosity Compensation Factor and Lohms) the actual power consumption will be only .10 HP (75 Watts) or about 20% less, while the lubricant flow will be 5% greater (greater lubricant flow increases engine component cooling and improves filtration rate).
This small decrease of .02 HP (15 Watts) in power required to drive the oil pump and to circulate the oil translates to about $81 in fuel savings over the 12 year life of the vehicle. Saving only $6.75 annually on fuel may not be very significant, but over the years it adds up and it helps to pay for the use of SynLube™ Lube‑4‑Life®
In large diesel truck and some racing engines the oil pump may consume as
much as 2 HP (1.5 kW) and therefore the use of SynLube™ Lube‑4‑Life® will contribute to a greater savings. Also keep in mind
that this power consumption is based on engine at normal operating temperature
and with oil sump temperature of 190°F (88°C), in cold engine with cold oil
the power consumption may be as much as eight times higher with
conventional petroleum motor oil, but only three times higher with SynLube™ Lube‑4‑Life® this is because SynLube™ Lube‑4‑Life® Lubricants are much less viscous and flow
easily at low temperatures down to -50°F (-45°C).
Petroleum motor oil, however, will be solid at about -30°F (-34°C).
Friction Losses in the engine account for about 10% to as much as 15% of the energy that is available in Gasoline. In really inefficient engine designs the Mechanical Friction Losses can be as much as 20%.
The Mechanical Friction Losses consist of following:

The above pie chart shows the distribution of the Mechanical Friction Losses in a typical engine.
The different concepts of friction and wear as well as the effect that SynLube™ Lube‑4‑Life® has on them is discussed in more detail on our web page SynLube MAGIC #2
|
|
 |
![]() Click the Left button or above heading for link to web page with Full Size animation of above.
Click the Left button or above heading for link to web page with Full Size animation of above.
You can see "How Engine
Works" animation videos on YouTube:
http://www.youtube.com/watch?v=_y6PS-2j2Ug
http://www.youtube.com/watch?v=IYWUWMmimjQ

Now that you understand how conventional Piston Engine works, it will be easier for you to comprehend the advantages and benefits that SynLube™ Lube‑4‑Life® offers.
To learn about them please see SynLube MAGIC section on our web site.